Cholecalciferol Injection IP
Cholecalciferol Injection IP Specification
- Best Before
- 24 months from date of manufacture
- Strength
- 600,000 IU/ml
- Shelf Life
- 24 months
- Flavor
- Unflavored
- Effect
- Corrects Vitamin D deficiency, improves calcium absorption
- Purity
- Above 98%
- Medicine Type
- Allopathic
- Efficacy
- High efficacy in treating and preventing Vitamin D deficiency
- Life Span
- Up to shelf life mentioned
- Brand Name
- As per packaging (varies with manufacturer)
- Chemical Composition
- Cholecalciferol 600,000 IU per ml
- Ingredients
- Cholecalciferol (Vitamin D3)
- Sub Ingredients
- Water for Injection, other inert excipients
- Function
- Antioxidant
- Dosage
- As directed by physician
- Dosage Guidelines
- With Water
- Dosage Form
- Liquid
- Storage Instructions
- Store in a cool and dry place, protect from light
- Packaging Size
- 1 ml ampoule
- Packaging Type
- Glass ampoule
- Warning
- To be used under medical supervision only
- Route of Administration
- Intramuscular injection
- Prescription/Non-prescription
- Prescription only
- Appearance
- Clear, colorless solution
- Indication
- Vitamin D deficiency, osteoporosis, rickets, osteomalacia
- Storage Temperature
- Below 25°C
- Contraindications
- Not to be used in hypercalcemia or hypersensitivity to vitamin D
- Sterility
- Sterile
Cholecalciferol Injection IP Trade Information
- Minimum Order Quantity
- 50 Boxes
- Payment Terms
- Western Union
- Supply Ability
- 1000 Boxes Per Month
- Delivery Time
- 7 Days
- Main Domestic Market
- All India
About Cholecalciferol Injection IP
Premium Features and Versatile Uses of Cholecalciferol Injection IP
Cholecalciferol Injection IP features a celebrated clear, sterile formulation, available as a 1 ml ampoule with a strength of 600,000 IU/ml. Used for correcting Vitamin D deficiency, managing osteoporosis, rickets, and osteomalacia, it is administered intramuscularly on prescription. Used as a potent antioxidant, this solution delivers rapid improvement in calcium absorption, aiding bone health. Its rarefied purity and high efficacy make it a favored choice among medical professionals for treating severe deficiencies.
Sample Policy, Certifications, and Main Export Markets for Cholecalciferol Injection IP
Request your Cholecalciferol Injection IP sample in accordance with our clear shipment and drop-off policy. Competitive list prices are available for qualified buyers. The injection is certified as per stringent quality standards, ensuring prime efficacy and sterility. Major export markets include Asia, Africa, and the Middle East, with distributors and traders prioritizing timely shipment and reliable customer support. Samples and bulk orders are processed efficiently for both domestic and export requirements.
FAQ's of Cholecalciferol Injection IP:
Q: How is Cholecalciferol Injection IP administered?
A: Cholecalciferol Injection IP is administered via intramuscular injection and should only be given under medical supervision as per dosing instructions provided by a healthcare professional.Q: What main conditions is Cholecalciferol Injection IP used for?
A: It is used primarily to treat and prevent Vitamin D deficiency, osteoporosis, rickets, and osteomalacia by improving calcium absorption and supporting bone health.Q: When should Cholecalciferol Injection IP not be used?
A: This injection should not be used in cases of hypercalcemia or in patients with hypersensitivity to vitamin D or any of the injection ingredients.Q: Where should Cholecalciferol Injection IP be stored?
A: Store the ampoules below 25C in a cool, dry place, protected from light, to maintain sterility and efficacy up to its 24-month shelf life.Q: What benefits can patients expect from using Cholecalciferol Injection IP?
A: Patients can expect improved correction of Vitamin D deficiency, enhanced calcium absorption, and support for bone strength and overall antioxidant protection.Q: What is the process for obtaining a sample of Cholecalciferol Injection IP?
A: Samples can be requested based on the distributor or supplier's policy; shipments and drop-offs are managed with transparency and in compliance with required certifications for major markets.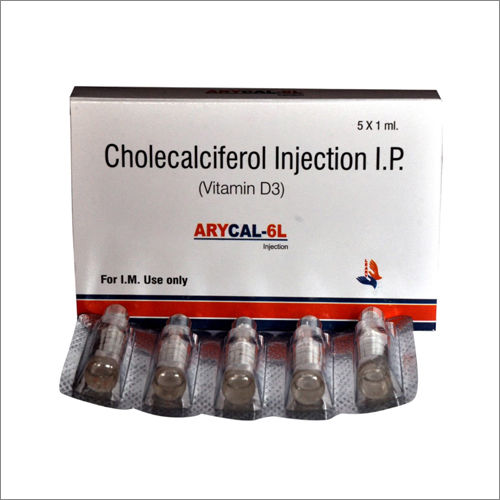

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Dietary Supplements Category
Bacillus Clausii Spores Suspension
Minimum Order Quantity : 50 Boxes
Storage Instructions : Store below 25C, Protect from direct sunlight
Purity : Sterile
Medicine Type : Allopathic
Sub Ingredients : Purified Water, Stabilizers, Preservatives
Chemical Composition : Each 5 ml contains Bacillus Clausii Spores (2 billion spores/5 ml)
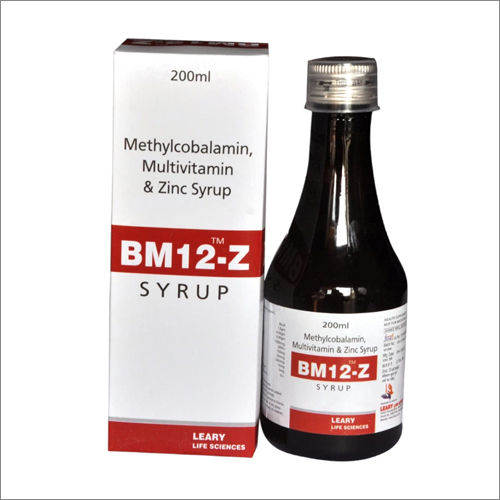
Methylcobalamin Multivitamin And Zinc Syrup
Minimum Order Quantity : 50 Boxes
Storage Instructions : Store in a cool, dry, and dark place away from direct sunlight
Purity : High
Medicine Type : Allopathic
Sub Ingredients : Vitamin B6, Vitamin B1, Vitamin B2, Folic Acid, Niacinamide, DPanthenol, Vitamin E, Vitamin C, Vitamin A
Chemical Composition : Methylcobalamin 1500mcg, Zinc 22.5mg, Multivitamins as per formulation

 Send Inquiry
Send Inquiry



 Send Inquiry
Send Inquiry Send SMS
Send SMS